BS Chemistry, 1979, Middle East Technical University, Ankara, Turkey
MS Organic Chemistry, 1981, Middle East Technical University, Ankara, Turkey
PhD Physical Organic Chemistry, 1986, Virginia Commonwealth University, Richmond, VA
Postdoctoral Fellow. Quantum Chemistry, 1986-89, University of Alabama at Birmingham, Birmingham, AL
Certificate, Executive Program for Scientists and Engineers, Business Management, 2001, University of California at San Diego, San Diego, CA,
My over 30 years of experience in computational chemistry started in 1982 with my PhD research that involved molecular orbital calculations to compare computational predictions of regio- and stereo-selectivity of some Diels-Alder reactions to experimental results. My postdoctoral fellowship that started in 1987 involved in quantum mechanical computational design of some high energy/density materials to be used in rocket propellants. From 1989 on my area of interest moved from materials science to life sciences dealing with chemical informatics systems (at MDL 1989-1996), molecular modeling technology (at Accelrys 1996-2005) to computer-aided drug design (at Turquoise Consulting 2006-2011, and at Mercer University 2012-2015). Following my retirement at August 2015, I am now employed, part-time, teaching chemistry at Santa Rosa Junior College.
Recent Publications:
2022:
Hughes, T.D; Güner, O.F.; Iradukunda, E.C.; Phillips, R.S.; Bowen, J.P. “The Kynurenine Pathway and Kynurenine 3-Monooxygenase Inhibitors," Molecules, 2022, 27(273), 1-26.
2020:
Uko, N.E.; Güner, O.F.; Matesic, D.F.; Bowen, J.P.; “Akt Pathway Inhibitors”, Current Topics in Medicinal Chemistry, 2020, 20, 1-18.
2019:
Uko, N.E.; Güner, O.F.; Bowen, J.P.; Matesic, D.F. “Akt Pathway Inhibition of the Solenopsin Analog, 2-Dodecylsulfanyl-1,-4,-5,-6-tetrahydropyrimidine”, Anticancer Research, 2019, 39, 5329-5338.
Murnane, K.S.; Güner, O.F.; Bowen, J.P; Rambacher, K.M.; Moniri, N.H,; Murphy, T.J.; Daphney, C.M.; Oppong-Damoah, A.; Rice. K.C., “The adrenergic receptor antagonist carvedilol interacts with serotonin 2A receptors both in vitro and in vivo”, Pharmacology, Biochemistry and Behavior, 2019,181, 37–45.
2018:
E. Blake Watkins, Osman F. Güner, Amol Kulkarni, Bernard Lassègue, Kathy K. Griendling and J. Phillip Bowen, "Discovery and Therapeutic Relevance of Small-Molecule NOX4 Inhibitors," Med. Chem. Rev., 2018, 53(8), 135-150.
Qian Xu, Amol A. Kulkarni, Ayyiliath M. Sajith, Dilbi Hussein, David Brown, Osman F. Güner, M. Damoder Reddy, E. Blake Watkins, Bernard Lassègue, Kathy K. Griendling, J. Phillip Bowen, "Design, synthesis, and biological evaluation of inhibitors of the NADPH oxidase, Nox4," Bioorg. Med. Chem. 2018, 26, 989-998.
Nne E. Uko, Osman F. Güner, Lillie M.A. Barnett, Diane F. Matesic, J. Phillip Bowen, "Discovery and biological activity of computer-assisted drug designed Akt pathway inhibitors," Bioorg. Med. Chem. Lett., 2018, 28, 3247-3250.
2017:
Robert S. Phillips, Andrew D. Anderson, Harvey G. Gentry, Osman F. Güner, J. Phillip Bowen, "Substrate and inhibitor specificity of kynurenine monooxygenase from Cytophaga hutchinsonii," Bioorg. Med. Chem. Lett., 2017, 27, 1705-1708
- Haizhen A. Zhong, Osman F. Güner, J. Phillip Bowen, "Rational Drug Discovery, Design, and Development," in Drug Delivery: Fundamentals & Applications, 2nd Ed. 2017, CRC Press, Boca Raton, FL. pp. 443-464
Patents:
- US 2020/0270214 A1 and WO 2019/023448 A1. Osman F. Güner, Bernard Lassegue, Kathy Griendling, Quian Xu, David Brown, J. Phillip Bowen, Amol Kulkarni, Blake E. Watkins, "NADPH Oxidase Inhibitors and Uses Thereof,"
2016 - to date Adjunct Faculty (teaching), Santa Rosa Junior College, Santa Rosa, CA
2012-2015 Research Scientist, Center for Drug Design, Mercer University, College of Pharmacy, Atlanta, GA
2006-2012 Principal, Turquoise Consulting, San Diego, CA, (2006-10), and Ankara, Turkey (2010-12)
1996-2005 Executive Director (last position), Accelrys, Inc., San Diego, CA
1989-1996 Senior Scientist (last position), MDL Information Systems, Inc., San Leandro, CA
1. GH-Score: Development of a metric to assess the quality of pharmacophore models and hitlists following database screening/searching. The GH-score (Goodness of Hit List, also recognized as the Güner-Henry score) is now utilized routinely for external validation of pharmacophore models and screening results.
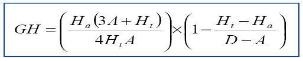
Following favorable results from a study that performed a comparative study of such metrics, GH-Score has become one of the most reliable external validations of search results.
“They found that the cumulative recall and the “Goodness of Hit List” or Güner-Henry (GH) score were among the most successful of those tested for measuring the effectiveness of similarity retrieval.”
Quoted from: Raymond, J.W. and Willett, P., J. Comp.-Aided Molec. Des., 2002, 16(1), 59-71
References:
- Güner, O. F. and Henry, D. R. "Metrics for Analyzing Hit Lists and Pharmacophores” in Pharmacophore Perception, Development, and Use for Drug Design, Güner, O. F. (Ed.), International University Line, 2000, 193-211
- Güner, O. F.; Waldman, M.; Hoffmann, R.; Kim, J-H., “Strategies in Database Mining and Pharmacophore Development,” in Pharmacophore Perception, Development, and Use for Drug Design, Güner, O. F. (Ed.), International University Line, 2000, 213-232
 2. First book on pharmacophores: The contributor/editor of the first book about pharmacophore modeling, entitled “Pharmacophore Perception, Development, and Use in Drug Design” published in 2000.
2. First book on pharmacophores: The contributor/editor of the first book about pharmacophore modeling, entitled “Pharmacophore Perception, Development, and Use in Drug Design” published in 2000.
Quoting Lisa Balbes in her book review for The Alchemist:
“In principle, it is a simple idea, but in practice the large number of different methods, as well as the wide variety of problems to which these methodologies are applied, make this a very hard field to get started in — much of the information is not published anywhere, and there is no single complete source that includes information on all the different methods. That is, until now. If I had to describe this book in one word, it would be ‘comprehensive’.”
References:
- Pharmacophore Perception, Development, and Use for Drug Design, Güner, O. F. (Ed.), International University Line, 2000, Ja Jolla California
- Güner, O. F. “History and Evolution of the Pharmacophore Concept in Computer-Aided Drug Design,” Curr. Top. In Med. Chem., 2002, 2, 1321-1332
- Güner, O. F.; Clement, O.; Kurogi, Y. “Pharmacophore Modeling and Three-Dimensional Database Searching for Drug Design Using Catalyst: Recent Advances,” Curr. Med. Chem., 2004, 11, 2991-3005
- Güner, O. F. “The Impact of Pharmacophores in Drug Design,” IDrugs 2005, 8(7), 657-672
3. Origin of the pharmacophore concept: The origin of the concept of pharmacophores has been recently challenged and incorrectly attributed to Lemont B. Kier based on his published model for muscarinic inhibitor compounds in 1967. In a recent paper, I demonstrated that similar models were published earlier by different authors, and following a detailed scientific detective work, have identified an 1898 paper of Paul Ehrlich as the originator of the concept.
Reference:
- Güner, O. F. and Bowen, J. P. “Setting the Record Straight: The Origin of the Pharmacophore Concept,” J. Chem. Inf. Model. 2014, 54, 1269-1283
Full list of publications up to 2006 can be accessed at Turquoise Consulting web site (http://www.turquoisecons.com/pubs.html ).
List of more recent publications can be accessed at ResearchGate (https://www.researchgate.net/profile/Osman_Guner/publications ).
Member, American Chemical Society (1985-2011)
ACS Chemical Information Division (1993-2011)
Program Committee Member, 1998-2001
Associate Program Chair, 2001-2003
Program Chair, 2003-2004
Voting member of the Executive Committee, 2003-2007
Chair-Elect, 2005
Finance and Fund Raising Committee member, 2005-2007
Chair, 2006
Awards Committee member, 2006-2008
Past-Chair, 2007
Nominations Chair, 2007
Fellow, American Institute of Chemists (1989)
Member, International QSAR and Modelling Society (2000-2010)
Member of the Board of Directors (2000-2010),
Trustee, Chemical Structure Association Trust (2006-2008)
Member of the Organizing Committee:
ADMET-1 Conference San Diego, CA, 2004
15th EuroQSAR 2004, Istanbul, Turkey, 2004
ADMET-2 Conference San Diego, CA, 2005